
Unify Your Life Sciences Operations in One AI-Powered Platform
Bring life-saving treatments to market faster by connecting your Regulatory, Safety, Quality, and Clinical teams in a single platform.
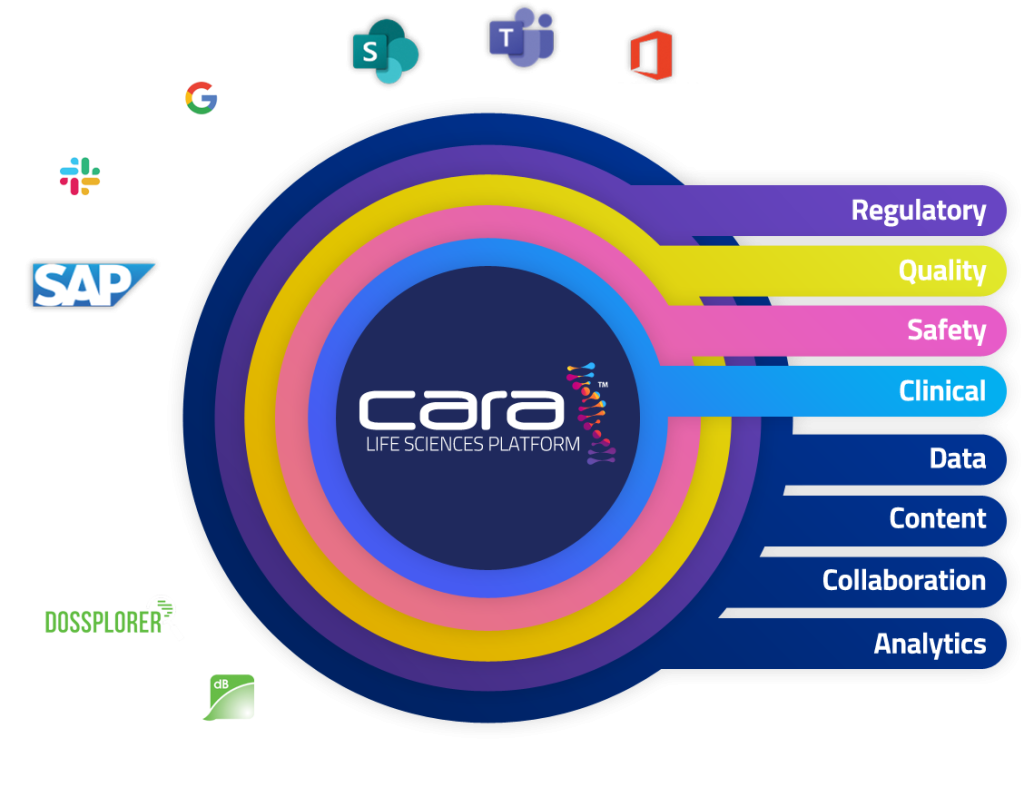












Transforming Regulatory Affairs
Simplify, standardise, and automate your regulatory processes with a data-driven platform.
Quality Reimagined
Gain control and understanding of your global processes to drive compliance.
Integrated Safety
Predict, analyse, and react faster with a seamless platform for safety data.
Clinical Without Boundaries
Communicate, adapt, and improve interoperability with a digital clinical platform.
The platform supports different, powerful functionality for each use case, e.g. security, versioning, workflows, lifecycles, tools, automations, and integrations.
Users who work across multiple parts of the business can have access to all use cases on the same platform
Data and content can flow between any solution on the platform without integration, enabling seamless collaboration, discovery, and traceability across the business.
The UI is common between all business processes, reducing training and improving adoption.
A True Platform
Data and content can flow between any solution on the platform without integration, enabling seamless collaboration, discovery, and traceability across the business.
Common Interface
The UI is common between all business processes, reducing training and improving adoption.
Powerful Process Automation
The platform supports different, powerful functionality for each use case, e.g. security, versioning, workflows, lifecycles, tools, automations, and integrations.
One User, One License
Users who work across multiple parts of the business can have access to all use cases on the same platform.


Efficiency gains in numbers
Building a Future
Instead of providing spot solutions that will be replaced in 5 years, the CARA Life Sciences Platform provides businesses with the foundational power to continuously innovate.
With immense flexibility and low-code rapid application building, Life Sciences companies can adapt, pivot, and implement new solutions & processes as they need to.
CARA provides not only the core best-of-breed applications that are industry standard, but also the ability to enable digital transformation of the processes that are specific to each business.
Building a Future
Instead of providing spot solutions that will be replaced in 5 years, the CARA Life Sciences Platform provides businesses with the foundational power to continuously innovate.

With immense flexibility and low-code rapid application building, Life Sciences companies can adapt, pivot, and implement new solutions & processes as they need to.
CARA provides not only the core best-of-breed applications that are industry standard, but also the ability to enable digital transformation of the processes that are specific to each business.

Latest Resources

Getting Value from AI : Why Life Sciences Companies Can’t Afford to Waste Time
Artificial intelligence is everywhere. Every week seems to bring another announcement promising smarter summarisation, faster drafting, or a new assistant that can shave seconds off routine tasks. For many organisations, these tools can be useful. They help individuals move faster, clear inboxes quicker, or generate a first draft more efficiently.

Turning Internal Knowledge into AI Intelligence: Why Context Is Everything
The Hidden Cost of Deploying AI Without Context There is a quiet assumption embedded in most conversations about AI adoption that deploying a large language model is, in itself, enough. Upload a document, ask a question, get an answer. The technology does the rest. This assumption is costing organisations more

The Prompt Problem: Why Uncontrolled AI Inputs Are a Compliance Risk
When AI Inputs Become a Compliance Liability When organisations evaluate AI tools, the conversation usually centres on model capability: accuracy, speed, hallucination rates. These matter. But they overlook a more operationally critical question: who controls the prompt? In generic AI platforms, prompt creation is left entirely to individual users. Everyone
Locations: Sarasota, FL • London, UK • Amsterdam, NL • Frankfurt, DE • Tokyo, JP

























